 
In addition, we have a molecular orbital theory (MOT), the most in-depth analysis of chemical bonding using quantum mechanical properties. 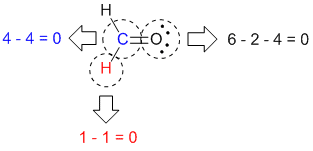
So far, we have covered many structural aspects of the formaldehyde molecule by using hybrid orbitals and VSEPR theory. For carbon, it is three.Īccording to the table below, for total domains (steric no.) = 3 and lone pair = 0, the molecular shape is trigonal planar.Īccording to VSEPR, the magnitude of repulsion increases in the order:īond pair- bond pair 120⁰ and the H-C-H bond angle closes to < 120⁰. of lone pair electrons on the central atom. of atoms bonded to the central atom plus the no. In formaldehyde, we will be considering the electron pairs around Carbon. The Valence Shell Electron Repulsion Theory attempts to predict the geometry of individual molecules using the concept of minimum energy and maximum stability.Īccording to VSEPR, the lowest energy can be achieved by minimizing repulsion between electron pairs around the central atom, giving the most stable geometry. Three hybrid orbitals can be formed from one s and two p orbitals giving sp2 hybrids. of valence electrons in the central atomįor formaldehyde, X = ½ * ( 2 + 4 + 0 + 0 ) = 3. Hybridization can also be calculated for a molecule using the formula We will see why the bond angles are not exactly 120⁰ in the next section. Oxygen is also sp2 hybridized in this molecule, but it forms only one σ bond as the other two sp2 hybrid orbitals are filled with lone pair electrons. It is used for sideways overlap to form a π bond with the 2p orbital of oxygen. The unhybridized 2p orbital on carbon is perpendicular to the molecular plane. The sp2 hybrid orbitals are planar with an angle of 120⁰. But there are three single bonds in Formaldehyde so hybridization becomes important. Unhybridized carbon will only be able to make 2 single bonds along the internuclear axis (usually the z-axis), using s orbital and one pz orbital. Carbon has an electronic ground state configuration of 1s2 2s2 2p2. In the case of formaldehyde, let’s take carbon. Hybridization helps us understand the nature of these covalent bonds using atomic orbitals of the central atom. Covalent bonds are directional which means they have a specific arrangement in space. We have the basic picture of bonding in the Lewis structure of H2CO but we still do not know about the shape of the molecule. So, the final Lewis structure, with zero formal charges is: Hydrogen: Formal charge = 1 – ½*2 – 0 = 0 Step 5: Now we calculate the formal charge on each atom,įormal charge=No. In this case, from oxygen, we draw in a lone pair to complete the octet around carbon. If there are no electrons left, a lone pair from a terminal atom can be used to form another bond. Step 4: We have to complete the octet on the central atom. That leaves 6 electrons, all of which are used upon oxygen. 
We have formed three bonds using 6 electrons for formaldehyde. It is a good idea to start with the most negative elements first while adding lone pair of electrons. Step 3: Every single bond has used up 2 electrons, so the rest are added as lone pairs to the terminal atoms to complete the octets, till there are no more electrons left. Now join other terminal atoms using single bonds to the central atom. In this case, it is carbon, which can form 4 bonds. of single bonds and/or able to expand its octet. It is usually the atom able to form maximum no. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
